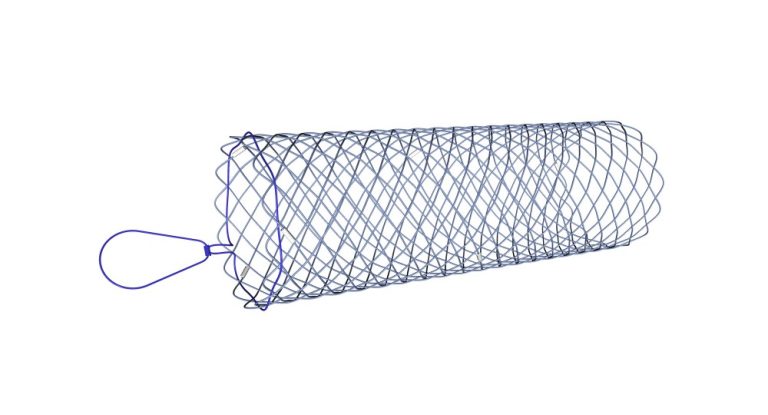
Self-expandable nitinol uncovered stent with high flexibility that allows implantation into anatomical curves. The atraumatic ends and expansion force of the stent provide ideal support for keeping the lumen of the pylorus or duodenum open.
SX-ELLA Stent Pyloroduodenal (Enterella) is indicated for palliative treatment of gastric outlet syndrome or duodenal strictures and/or obstructions caused by malignant tumors.
The delivery system with braided catheter allows an easy passage and is kink-resistant.
The atraumatic ends of the stent act to prevent injuries to the duodenal mucosa.
The stent is made of high quality nitinol. The radiopaque markers made of platinum-iridium alloy located at both ends and in the middle of the stent allow good visibility and accurate stent placement.
The plastic retrieval fiber allows repositioning up to 5 days after implantation.
MRI compatibility - "MR Conditional", compatible with 1.5 Tesla and 3 Tesla static magnetic field.
The stent can be inserted endoscopically and/or fluoroscopically.
An endoscope with a working channel of 4.2 mm is suitable for stent insertion.
The stent is delivered sterile, compressed in a delivery system. A 0.035 ”(0.89 mm) / 450 cm stiff guidewire is used for insertion.