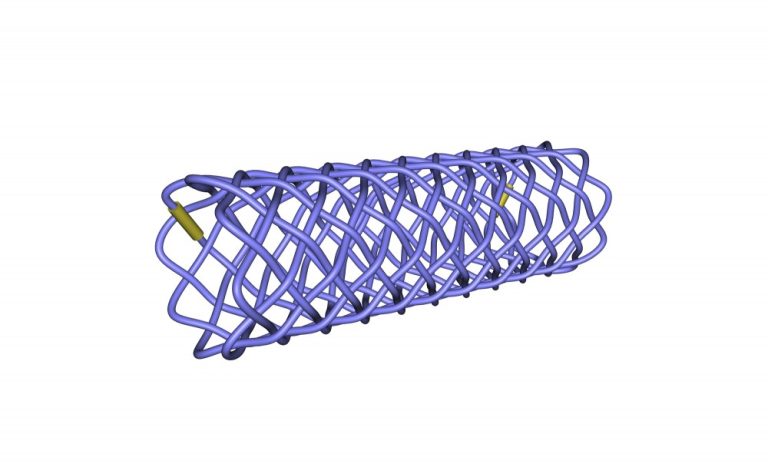
The DV Stent Biliary THP is a worldwide unique self-expandable biliary biodegradable stent that degrades in the human body usually 3 to 6 months after the implantation. Therefore, it is an ideal solution for temporary use in benign indications, where it is not necessary to extract it from the body like other non-degradable stents.
The DV Stent Biliary THP is intended for patients aged 18 and older and is indicated for the treatment of benign biliary stenoses.
The DV Stent Biliary THP degrades in the human body usually in 3 to 6 months. This reduces the number of interventions and hospital visits.
The stent is made of polydioxanone, an absorbable material (polymer) used in medicine as a surgical suture for more than 30 years.
The stent integrity and radial force are maintained for 6 to 8 weeks after implantation. Subsequently, the radial force gradually decreases until the degradation of the material after 3 to 6 months.
The DV Stent Biliary THP eliminates the need for repeated exchange of a biliary catheter and extraction of non-degradable stents which decreases the risk of infection or other post-procedural complications.
Sufficient radial force to maintain the bile duct lumen open in benign strictures.
The pH value affects the stent degradation process. At higher pH, stent degradation is slower, at lower pH, stent degradation is faster.
Low migration is achieved by uncovered stent design.
Polydioxanone is not visible under X-ray, therefore the stent is equipped with gold markers - 1 piece at both stent ends.
The delivery system with braided sheath is kink-resistant. The atraumatic olive and the radiopaque ring at the end of the delivery system allow easy insertion. The delivery system also has the option of retracting the partially released stent during implantation, up to 60% of the stent length.
MRI compatibility - "MR Conditional", compatible with 1.5 Tesla and 3 Tesla static magnetic field.
The stent is supplied sterile and is packed separately from the delivery system and the compression tool (ie, a tool for easier compression of the stent into the delivery system). The stent is inserted percutaneously under fluoroscopic control. Just prior to implantation, the stent has to be manually compressed into the delivery system. We recommend using a 0.035 ”(0.89 mm) / 180 cm ultra stiff guidewire for the implantation.